This website is intended for healthcare professionals only.
Take a look at a selection of our recent media coverage:
4th March 2024
While a significant proportion of patients with myocardial infarction are prescribed clopidogrel, common genetic variants mean that some people – and more from certain ancestry groups – may not see its preventative benefit.
Here, Dr Emma Magavern, clinical research fellow for the Centre of Clinical Pharmacology and Precision Medicine at William Harvey Research Institute, Queen Mary University of London, UK, discusses her most recent findings and the value of clinicians adopting pre-emptive pharmacogenomic testing.
As clinicians, we know that there is inter-individual variability in medication response. Different people respond differently to the same medication; some take medication and do not experience the intended benefit, and some people unfortunately experience adverse drug reactions (ADRs). Some of this variability in response is due to genetics.
We already use other information known to impact on risk of ADRs from a medication in our clinical practice. Examples include checking liver and renal function before prescribing medicines.
By using information on very common genetic markers to inform prescribing, we can optimise the benefits of medications and minimise the risk of side effects. This area, known as pharmacogenomics, has however been slow to enter mainstream clinical practice.
The recently published PREPARE trial showed that a pre-emptive pharmacogenomic testing approach, where the genotype informs prescribing, can decrease ADRs by one-third in a multi-centre prospective clinical trial setting.1
ADRs pose significant problems for patients and take a large toll on healthcare systems. Studies estimate ADRs are responsible for 6.5% of hospital admissions in the UK and cost more than £2bn a year to the NHS.2,3 So there is clearly much room for improvement.
People from certain ancestry groups may have a higher risk of experiencing a side effect or not benefiting from medication due to common genetic variants. An example is illustrated by the use of clopidogrel after a myocardial infarction.
Clopidogrel is a commonly prescribed P2Y12 inhibitor used for secondary prevention after an initial ischaemic event.4 It is a prodrug and must therefore be activated in the body, via metabolism by cytochrome P450 2C19 (CYP2C19) in the liver, to achieve potency. Studies have shown that 60–70% of people who suffer from a myocardial infarction are prescribed clopidogrel.5
The CYP2C19 enzyme is encoded by the CYP2C19 gene, which is highly polymorphic.6 People with CYP2C19 loss of function (LOF) variants have higher on-treatment platelet reactivity with clopidogrel and are at a greater risk of secondary ischaemic events.7
LOF variants in CYP2C19 are common – present in approximately one in three people of European ancestry.7 Therefore, genetic variants resulting in individuals not being able to activate clopidogrel are common.
However, these variants are even more common in certain ancestral groups. Genetic changes that lead to poor activation of clopidogrel are most common in Asian and Oceanic ancestry groups.8
So, these ancestry patients have a higher risk of clopidogrel being ineffective than European ancestry participants, in whom antiplatelets were predominantly trialled, and therefore might benefit most from the use of genetics to help determine clopidogrel safety and efficacy (see Table 1 below).
Few studies have linked genetic data from under-represented populations with medication exposure and health outcome data. The only study with a substantial Asian population was focused on stroke rather than myocardial ischaemia and undertaken in east Asia.
Table 1. Studies supporting clopidogrel licensure as listed in the European Medicines Agency summary of product characteristics9–16
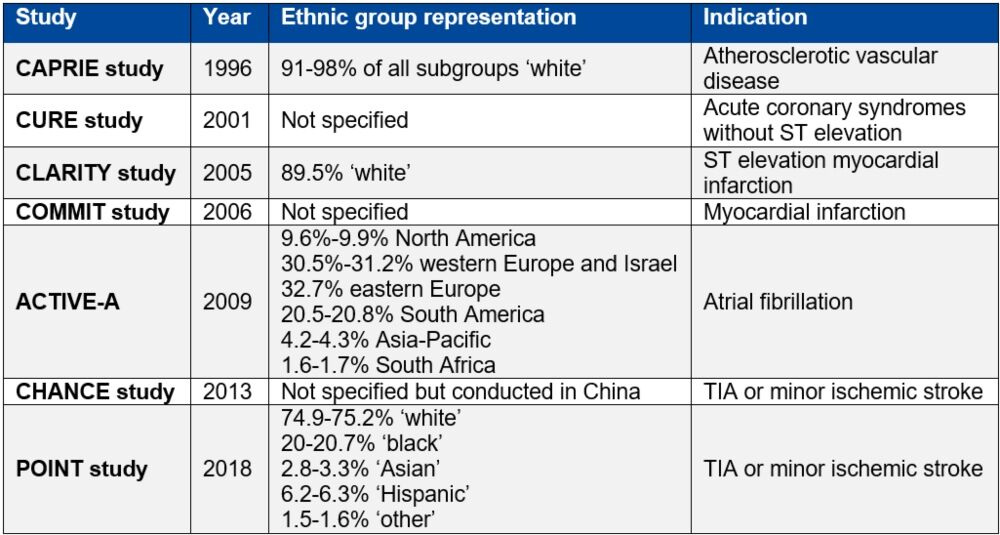
Table reproduced from Magavern E et al. JACC Adv 2023;2(7):100573. Copyright © 2023, The Authors. Open Access Article under the CC BY License.
The British-Bangladeshi and British-Pakistani populations in the UK are known to experience high rates of cardiovascular disease. The Genes & Health study recruited more than 50,000 people from this population and linked genotype data with national electronic health records data and primary care prescription records.17
In an analysis of 44,396 participants, we found that 57% of people in this group had a CYP2C19 LOF variant.18 Of these, 13% had two copies of an LOF variant – one from each parent (Figure 1).
Figure 1. Prevalence of CYP2C19 LOF alleles in the Genes & Health study’s South-Asian ancestry population
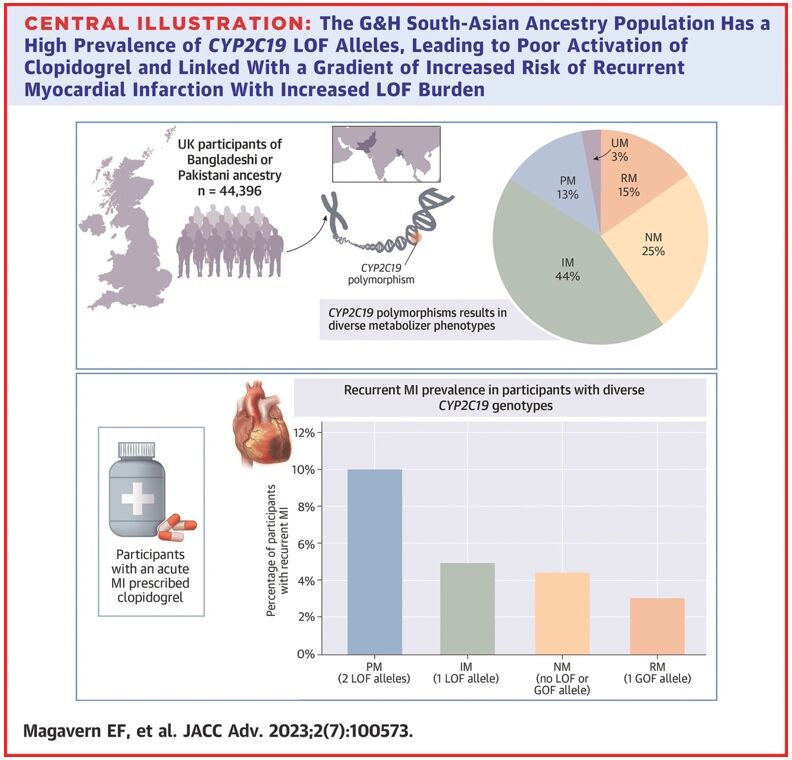
Reproduced from Magavern E et al. JACC Adv 2023;2(7):100573. Copyright © 2023, The Authors. Open Access Article under the CC BY License.
We identified people in this group who had experienced a myocardial infarction and were prescribed clopidogrel by their GP (697 participants). Those with a recurrent myocardial infarction were almost four-times more likely to have two LOF CYP2C19 alleles as compared with none. A total of 69% of participants who had a myocardial infarction were prescribed clopidogrel.18
This study showed how common genetic resistance to clopidogrel is in this population. It also demonstrated the widespread use of clopidogrel and linked genetic resistance with a higher risk of recurrent myocardial infarction in this population for the first time, using real-world data.
While a precision prescribing approach using CYP2C19 testing would benefit patients from all ancestries, it would disproportionately benefit the South-Asian ancestry population due to the higher prevalence of both coronary artery disease and genetic resistance to clopidogrel in this population.
Genetic testing may soon be available within the NHS to determine if stroke patients would benefit from clopidogrel.19 This means that some patients might already know their CYP2C19 genotype when they present with a secondary ischaemic event.
This information could subsequently be used to administer the most effective medicine and reduce their risk of further ischaemic events.
For pharmacogenetic testing to benefit patients who have had a myocardial infarction, several things must occur:
There should also be discussion with the public around this use of genetic testing, to raise awareness and design clinical pathways fit for purpose. These dialogues must include historically under-represented ancestry groups and build trust.
The goal is to make genetic testing possible in routine clinical care to help optimise antiplatelet choice after an ischaemic event and to ensure better outcomes for all, with a particular focus on improving health equality for the British South-Asian ancestry population.
Emma Magavern MD MSc MRCP
Clinical Research Fellow, Centre of Clinical Pharmacology and Precision Medicine, William Harvey Research Institute, Queen Mary University of London, UK
26th May 2023
A genetic ‘biobank’ will be launched on 1 June 2023 by the UK’s Medicines and Healthcare products Regulatory Agency (MHRA) to understand how a patient’s genetic make up can impact the safety of their medicines.
Known as the Yellow Card biobank, the biobank will contain genetic data and patient samples and be used by scientists to determine whether a side effect from a medicine was caused by a specific genetic trait.
A joint venture with Genomics England, the genetic research resource is the first of its kind to be launched by a drug safety regulator.
Operating alongside the MHRA’s existing Yellow Card reporting site for suspected side effects and adverse incidents involving medicines and medical devices, the new venture forms part of a long-term vision for more personalised medicine approaches.
By understanding the underlying mechanism of an adverse drug reaction (ADR), it is hoped that pharmacogenetic testing strategies could be developed so that ADRs could be prevented rather than requiring a reactive approach.
In turn, this will enable doctors to target prescriptions so UK patients will receive the safest medication for them, based on their genetic makeup.
‘We are excited by the upcoming launch of the Yellow Card biobank, which demonstrates that we are at the absolute forefront of innovation in the field of drug safety monitoring,’ said Dr June Raine, MHRA Chief Executive. ‘Almost a third of adverse reactions to medicines could be prevented with the introduction of genetic testing… This has the potential to transform our safety monitoring activities – enabling us to meet a real need by using high-quality patient data to reduce side effects of medicines.’
Recruitment for the biobank will commence on 1 September and participants will be visited at home by a nurse who will take a blood sample to be added to the biobank. The sequencing of participants’ genetic material will begin in spring 2024. Initial research findings are due to be published in 2025.
The pilot phase will start by looking at allopurinol and related rare, severe skin reactions including Stevens-Johnson syndrome and toxic epidermal necrolysis. Other topics of focus for the pilot phase will be confirmed in due course.
Genomics England will support the MHRA with the sequencing and storage of genetic material through use of its well-established and secure infrastructure.
Commenting on the ‘transformative partnership’ with the MHRA, Professor Matt Brown, Chief Scientific Officer for Genomics England, added: ‘Many [severe ADRs] are influenced by underlying genetic risk factors, substantially heightening an individual’s vulnerability.
‘By joining forces with the MHRA, we are poised to gain greater understanding of these genetic influences – discoveries that will be vital if we are to move to harness the power of genomics to proactively protect patients from these harms.
‘Together, we hope that this is the first step towards redefining the future of drug safety.’
ADRs continue to be a significant burden on the NHS and account for one in 16 hospital admissions.