This website is intended for healthcare professionals only.
Take a look at a selection of our recent media coverage:
22nd May 2024
Penicillin allergy is a common concern in healthcare settings, impacting patient safety, antimicrobial stewardship and successful infection treatment. Gerry Hughes explores the epidemiology of penicillin allergy and its significance in the context of patient care, as well as the need for a system-wide and multidisciplinary approach to penicillin allergy delabelling.
Penicillin is commonly implicated in drug hypersensitivity. In developed countries, between five and 15% patients carry a penicillin allergy label, and patients receive a penicillin allergy label by their third birthday in approximately 75% of cases.
However, a large body of evidence suggests upwards of 90% of patients with a penicillin allergy label are not truly allergic. In England alone, 5.9% of the population are designated penicillin allergic, with an estimated 2.7 million of these being incorrectly labelled as such.
It is important for patients and patient carers to know that common antibiotic adverse events, such as upset stomach, vomiting or diarrhoea are not symptoms of penicillin allergy. The Gell and Coombes classification of drug hypersensitivity (see Table 1 below) provides a useful reference for assessing and diagnosing drug-induced allergic reactions.
Hypersensitivity reactions associated with penicillin are predominantly Type I (anaphylactic, immediate onset) or Type IV (cell-mediated, delayed skin reactions).
Given the prevalence and implications of incorrect penicillin allergy labels, it is important for healthcare professionals to understand the impact of this issue on the successful treatment of infections and the broader implications for antimicrobial stewardship (AMS).
Table 1: Gell and Coombes hypersensitivity classification*
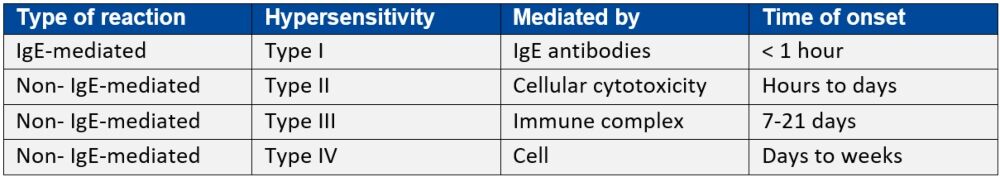
IgE: immunoglobulin E
*Adapted from Penicillin allergy: A practical guide for clinicians
Inappropriate labelling of penicillin allergy negatively impacts on patient care for several reasons. For many infections, penicillin, or beta-lactam antibiotics, are first choice therapy due to the weight of evidence behind their effectiveness.
Penicillin avoidance, due to a spurious penicillin allergy designation, can lead to use of less effective antibiotic regimens, unnecessary exposure to alternative antibiotic therapy, development of Clostridium difficile infection, additional time required in hospital and increased overall care costs.
The UK Health Security Agency’s ‘Start smart then focus’ AMS toolkit – first published in 2011 and most recently updated in September 2023 – makes evidence-based recommendations on effective AMS practices in hospitals, including appropriate management of penicillin allergy labels.
The toolkit advises secondary care clinicians and leaders about how local AMS policies and guidelines should include assessment of reported drug allergies and encourages them to conduct allergy delabelling where possible ‘to ensure patients are not denied access to the most effective therapy’.
Penicillin allergy assessment and delabelling (PAD) seeks to investigate a penicillin allergy history, remove the allergy designation where possible and/or desensitise the patient to future penicillin therapies.
Although immunology specialists are well-suited to develop and conduct PAD, it is a role often lacking in hospitals and other clinical environments. The British Society for Allergy and Clinical Immunology states that PAD performed by allergy specialists only, ‘cannot meet either current or future demand’.
Indeed, a recent study published in the Journal of Infection suggested that inaccurate penicillin allergy labels are magnified by insufficient allergy specialists. It investigated the feasibility of a direct oral penicillin challenge in delabelling low-risk patients with penicillin allergy by non-allergy healthcare professionals.
PAD is an evidence-based component of AMS and has previously demonstrated benefits in optimising antimicrobial choice and allowing for cost-effective treatment choices.
This evidence base extends to a multidisciplinary team (MDT) approach to PAD, which harnesses the expertise of infection specialists from medicine, pharmacy and nursing, amongst others.
An excellent example of this MDT strategy is the PAD toolkit developed by the Scottish Antimicrobial Prescribing Group (SAPG). This suite of resources is for non-allergy specialists to aid removal of penicillin allergy labels from patients with unverified allergic reactions, including resources for primary care practitioners and patients.
Professor Andrew Seaton, consultant in infectious diseases and general medicine at NHS Greater Glasgow and Clyde in Scotland, is chair of SAPG and recognises the importance of these guidelines in practice.
‘Whilst removing an allergy label sounds easy to do, it is difficult to do safely and sustainably without a formal structure,’ he says. ‘The SAPG toolkit has been a collaborative, multidisciplinary development which provides that structure and assurance for both clinicians and patients.’
St James’s Hospital (SJH) in Dublin, Ireland, provides such MDT-led PAD services, led by Professor Niall Conlon, consultant clinical immunologist and professor of clinical immunology at Trinity College Dublin.
Echoing Professor Seaton’s comments, Professor Conlon notes that successful PAD programmes require a systems-level approach. He underlines the need for an adequately resourced service, underpinned by commensurate education and training for the MDT involved. ‘A cohesive strategy is needed where all clinicians, not just allergy specialists, feel comfortable performing delabelling,’ he says.
In line with SAPG recommendations, SJH also provides resources to patients on penicillin allergy, including information on confirmed penicillin allergy.
Neil Powell, consultant antimicrobial pharmacist and associate director, antimicrobial stewardship at Royal Cornwall Hospitals NHS Trust in England, is the recipient of a clinical academic grant from the National Institute for Health and Care Research.
His research work focuses on removing erroneous penicillin allergy labels and leveraging an MDT approach to achieve that. He is currently developing a complex intervention that will facilitate PAD to be delivered by ward pharmacists and ward doctors.
Describing his work, he says: ‘This will enable penicillin allergy delabelling to be delivered to more patients across the hospital, with appropriate governance frameworks to ensure it is done safely.’
As part of Mr Powell’s research, a recent qualitative assessment of PAD facilitators and barriers found broad support among healthcare workers for an MDT approach. Driven by a desire to ensure patient safety, participants felt that PAD should be a multidisciplinary responsibility, shared between doctors and pharmacists and supported by pharmacy technicians and nurses.
However, the study also found that patient engagement and education on the topic was key. As with all healthcare interventions, participants also recognised the need for an evidence-based service, with sufficient resources to support its aims and objectives.
Mr Powell says: ‘From the qualitative studies I have done, healthcare workers have said that it needs to be a shared responsibility, not the responsibility of one specialty.’ In particular, he notes that ward pharmacists are well placed, as experts in medication safety and medication optimisation, to support and deliver PAD.
In fact, in September 2023, the Royal Pharmaceutical Society (RPS) launched a new checklist for pharmacists and pharmacy teams to help inform conversations with patients about penicillin allergy and determine their true status.
The checklist states that on admission to hospital pharmacy professionals should ensure allergy history is reviewed as part of the drug history and medicines reconciliation process. It also refers to guidance on setting up non-specialist allergy delabelling services in hospitals from the British Society of Allergy and Clinical Immunology.
PAD may also have a place beyond secondary care settings, and this goes further than simply providing education to primary care practitioners.
The RPS checklist, for instance, encourages GP practice pharmacy teams can run searches to identify any patients with a documented allergy that have since received penicillin and ensure their records are updated to reflect this.
The ALlergy AntiBiotics And Microbial resistAnce (ALABAMA) trial, based in the UK, aims to determine if a primary care penicillin allergy assessment package is safe and effective in improving patient health outcomes and antibiotic prescribing.
This multicentre, parallel-arm, open-label, randomised pragmatic trial aimed to recruit between 656 and 848 participants from participating NHS general practices in England. Recruitment was completed in 2023 and results are expected this coming autumn.
Recognising the potential to expand PAD to community settings, Professor Seaton highlights the continued need for collaboration. ‘If we can scale up [PAD] we can expand initiatives beyond secondary care and into the community. Undoubtedly a multidisciplinary approach is needed, [and] involvement of clinical pharmacists and specialist nurses will be crucial.’
It’s clear that penicillin allergy delabelling is an essential component of AMS, with important implications for patient care and public health. More work is required to educate healthcare professionals across sectors, as well as the public, on the prevalence and consequences of inaccurate penicillin allergy labels, but work is underway to support this across the UK and in Europe.
Ultimately, development of well-resourced MDTs and evidence-based guidelines will facilitate a safe and effective delabelling process, ensuring that patients receive the most appropriate treatment for their condition, all while minimising the risk of adverse reactions and keeping patients safe.
26th March 2024
It is likely to be safe for penicillin allergy labels (PALs) to be removed for most patients, a study from the University of Birmingham has found.
Patients who thought they had an allergy to penicillin and who were deemed low risk took part in ‘penicillin challenges’, where a small dose of penicillin was given instead of allergy testing. Some 97% of participants labelled with PALs were shown to have no allergy.
The findings, published in the Journal of Infection, could improve care for individuals by giving patients a wider choice of antibiotics and easing the burden of antimicrobial resistance as more patients can safely be given penicillin.
A total of 6% of the population in England is labelled with a PAL, and 15-20% of patients in secondary care have a stated penicillin allergy. However, a large body of evidence suggests that 90-95% of PALs are inaccurate. Within the NHS, there is no point-of-care test for penicillin allergy, and tests are onerous and time-consuming.
Researchers and clinicians worked across three UK hospital trusts, University Hospitals Birmingham NHS Foundation Trust (UHB), Leeds Teaching Hospitals NHS Trust and Oxford University Hospitals NHS Foundation Trust, to deliver the penicillin challenges.
Over 2,000 patients were initially screened for eligibility in the study and categorised as low- or high-risk. Across the three specialties of acute medical, pre-surgical and haematology-oncology, 126 low-risk patients took part in the study.
Participants received an oral dose of amoxicillin, delivered by a research nurse or research pharmacist supervised by a non-allergy specialist clinical consultant. Resuscitation facilities were available if needed.
Of the 126 participants, 97% had no penicillin allergy, and no serious hypersensitive reactions were reported. The researchers showed how a closely monitored protocol for taking penicillin directly, rather than using a skin allergy test that needs to be delivered by an allergy specialist, was effective in low-risk patients, allowing these patients to safely use penicillin in the future.
Professor Mamidipudi Thirumala Krishna, chair of Allergy, Clinical Immunology and Global Health at the University of Birmingham and chief investigator of the study, said that inaccurate penicillin allergy labels are a huge global burden.
He explained: ‘Penicillin allergy labels are not benign and contribute to antimicrobial resistance, so enabling more patients to safely benefit from penicillin will ease the burden of other antibiotics that are currently being overused and improve quality of clinical care.’
The researchers state that inaccurate penicillin allergy labels are magnified by insufficient allergy specialists and a lack of point-of-care tests. Inaccurate penicillin labelling constitutes a major public health challenge, but this study could provide a low-cost framework that healthcare systems can adopt.
Dr Louise Savic, consultant anaesthetist and drug allergy specialist at Leeds Teaching Hospitals NHS Trust, and co-chief investigator, added: ‘This study demonstrates that a routine programme of de-labelling people who believe they are allergic penicillin, outside the setting of a specialist allergy clinic, is potentially achievable.
De-labelling was particularly successful within the outpatient population, suggesting that future efforts might be best targeted to this group in order to maximise benefit.’
This study follows research published in 2023 which found the use of a direct oral penicillin challenge in patients with a low-risk of penicillin allergy was non-inferior to the standard-of-care skin testing.
A version of this article was originally published by our sister publication Nursing in Practice.
18th July 2023
Use of a direct oral penicillin challenge in patients with a low-risk of penicillin allergy has been shown to be non-inferior to the standard-of-care skin testing in the recent PALACE randomised trial.
Removing the label of being allergic to penicillin involves intra-dermal skin testing followed by an oral penicillin challenge. Now, an international research group has shown that a direct oral penicillin challenge in those with a low-risk penicillin allergy is equally as safe as the current standard-of-care assessment.
In the parallel, two-arm, non-inferiority, open-label, randomised trial, patients with a PEN-FAST score below three, were randomly allocated to either the intervention group who had a direct oral challenge with penicillin, or the control group who received the standard of care.
The primary outcome of interest was a physician-verified positive immune-mediated oral penicillin challenge within one hour after the intervention. Researchers set the non-inferiority margin as a risk difference of less than five on a one-sided 95% confidence interval.
The trial recruited a total of 382 adults who were randomised, although only 377 patients with a median age of 51 years (65.5% female) were included in the final analysis. Among these, 187 were allocated to the intervention group. Most patients had a PEN-FAST score of 0 or 1.
The primary outcome occurred in only a single patient in both groups, giving a risk difference of 0.0084, which was below the non-inferiority margin.
In the five days following the oral penicillin challenge, a total of nine immune-mediated adverse events were recorded in the intervention group and 10 in the control group. None of these were deemed to be serious.
Dr Ana-Maria Copaescu, first author of the study and associate investigator in the Infectious Diseases and Immunity in Global Health Program at the Research Institute of the McGill University Health Centre in Quebec, said: ‘The biggest takeaway from the PALACE study is that patients with a low-risk penicillin allergy, like a childhood rash, can safely have a test dose of penicillin to determine if they are still allergic.
‘This will change the way doctors test for penicillin allergy in the future. Millions of patients worldwide… will be able to have their penicillin allergy disproved by a safe single oral test dose following a carefully risk-validated risk assessment.‘
Antibiotics such as penicillin are widely used in clinical practice although a true allergy to these drugs is rare with an estimated frequency of anaphylaxis at one to five per 10,000 cases. Hypersensitivity reactions are significantly more common, resulting in symptoms such as nausea, vomiting, pruritus, urticaria and wheezing.