This website is intended for healthcare professionals only.
Take a look at a selection of our recent media coverage:
11th September 2023
Retinal thinning appears to a consistent finding in patients with Parkinson‘s disease and questions have arisen over whether this serves as an early biomarker for the subsequent onset of the disease. Clinical writer Rod Tucker delves into the evidence.
Parkinson’s disease (PD) is estimated to affect at least 1% of people over the age of 60. The neurodegenerative condition affects both movement and cognition and is linked with the loss of dopaminergic neurons in the substantia nigra together with the presence of Lewy bodies.
Symptoms and progression of PD depend on the subtype, but patients with PD commonly experience tremor, bradykinesia and muscle stiffness, and up to 70% also have recurrent visual complaints and visual hallucinations.
But what is responsible for the visual dysfunction, and is it somehow linked to direct dopaminergic impairment within the eyes?
The fact that dopaminergic neurotransmission occurs within retina has been known since 1982, and the first evidence that patients with PD could experience visual problems emerged in 1987. Researchers observed a loss in both flicker sensitivity and near the peak of the spatial contrast sensitivity in those with the disease. But whether these visual deficits was causally related to PD remained unknown.
Definitive proof linking visual dysfunction with PD finally arrived, but from a rather unusual source. Canadian researchers decided to measure the level of dopamine in the retinas of eight patients with PD who died.
They stratified their findings based on each individual‘s last dose of levodopa therapy. While the retinal dopamine content in five patients who received levodopa therapy before death was similar to that in the controls, the levels were significantly lower in those who had not received the drug within the last five days before death.
As it became widely acknowledged that retinal levels of dopamine were often reduced in patients with PD, whether this was also associated with a concurrent loss of retinal tissue was less clear. Establishing such a relationship became much easier in 1994, following the introduction of optical coherence tomography (OCT).
This technique, which uses near-infrared light, enabled an assessment of nerve fibre thickness in the eye. In 2004, using OCT, researchers were able to show there was reduced thickness of the circumpapillary retinal nerve fibre layer in patients with PD.
Later work quantified this reduction in comparison to healthy controls and established that retinal thinning of the inner retinal layer was significantly greater in those with PD.
Although there was an emerging consensus that retinal thinning was a hallmark of PD, it wasn‘t actually a universal finding. For instance, other work was unable to detect a difference in comparison to healthy controls. A second study, which also failed to detect differences with PD and healthy controls, called for further work to define the role of OCT as a diagnostic biomarker.
Nevertheless, in a 2018 study, Korean researchers not only re-affirmed the presence of retinal thinning in patients with PD but provided the first evidence that there was a correlation between retinal thinning and nigral dopaminergic neuronal loss within the brains of patients who were at an early stage of their disease.
The relationship between retinal changes and PD was further strengthened in a 2021 meta-analysis of 27 studies that included 1,470 patients. The authors concluded that whole thickness – the thickness of the combination of ganglion cell layer and inner plexiform layer – and nerve fibre layer of central retina are significantly reduced in PD patients.
With confirmation of the relationship between retinal thinning and nigral dopaminergic neuronal loss, researchers sought to address a further pertinent question. If retinal thinning could be detectable in those without overt PD, could it serve as a prognostic marker for the disease?
This was the question posed in a recent study in the journal Neurology. Researchers assessed retinal thinning in two separate cohorts: one in patients with established PD and one in a second prospective cohort where retinal imaging had been performed.
The team looked at several aspects of retinal tissue including the ganglion cell-inner plexiform layer (GCIPL) and inner nuclear layer (INL) thicknesses, which had been measured in the two cohorts with OCT.
As expected, individuals in the first cohort with prevalent PD had thinner GCIPL and INL when compared to healthy controls. But what was more intriguing was how, in the prospective cohort in which patients had undergone OCT imaging approximately 2,653 days before the onset of PD, there was also significant GCIPL and INR thinning in those who went on to develop PD.
For example, the hazard ratio for a thinner GCIPL was 0.62 per standard deviation increase. In other words, for each standard deviation increase in GCIPL thickness, the risk of developing PD was reduced by 38%. In fact, the relationship remained significant even after excluding patients who developed PD within the first 24 months after their retinal imaging.
Taken together, the findings from the current study suggest that retinal thinning can be detected almost seven years before the onset of PD.
With the recognition that visual dysfunction in patients with PD is a harbinger of cognitive decline and greater white matter damage over time, it seems that the retina could become a potential imaging target.
There is still much more work that needs to be done, in particular, establishing the sensitivity and specificity of these changes, before the results can be used to predict whether an individual will develop PD. But if retinal thinning does turn out to be an effective biomarker, it could be used to support the early diagnosis of PD, thereby allowing for more effective treatment planning in those deemed to be at risk of developing the condition.
Guidance on how global warming can be addressed in clinical practice has been outlined by the European Respiratory Society (ERS) in its latest consensus statement on climate change and respiratory health.
Published in the European Respiratory Journal, the statement describes climate change as ‘an unfolding major planetary and health crisis’, and a major threat to those with common lung conditions.
This, it says, is linked to the frequent and extreme weather events, prolonged aeroallergen seasons and poorer air quality associated with climate change, which can lead directly to a worsening of health and an increased risk of death.
Traditionally, clinicians have been involved in climate change adaptation strategies such as identifying vulnerable groups and providing advice on how they can protect themselves during heatwaves, for example.
However, the ERS says this clinical role has now expanded to focus on both human and planetary health, which includes contributing to the reduction in greenhouse gas emissions.
According to the ERS, subsequent changes to clinical practice could therefore include promoting green prescriptions such as inhalers; focusing efforts on smoking eradication; and encouraging patients, where appropriate, to engage with nature, take active modes of transport and make more sustainable food choices.
Professor Zorana Jovanovic Andersen, chair of the ERS Environment and Health Committee and professor of environmental epidemiology at the University of Copenhagen, who was one of the authors, said: ‘As respiratory doctors and nurses, we need to be aware of these new risks and do all we can to help alleviate patients’ suffering. We also need to explain the risks to our patients so they can protect themselves from adverse effects of climate change.‘
The consensus statement also highlights that climate change will have a disproportionately greater adverse effect on individuals living with respiratory diseases such as asthma and chronic obstructive pulmonary disease (COPD).
It outlines some of the health risks of climate change such as a decline in lung function, increases in allergic responses and/or new cases of chronic (asthma, COPD, lung cancer) or infectious (pneumonia, influenza, tuberculosis, Covid-19) respiratory diseases.
It also identifies a higher risk of exacerbations for existing respiratory diseases, increased use of medication, emergency department visits, hospitalisations and death.
The fact that children are more susceptible to the impact of climate change on lung health is also highlighted, including the fact that the prevention of chronic respiratory disease should start as early as possible as many chronic lung diseases in adults have childhood origins.
Several previous reviews have provided extensive summaries of the different mechanisms by which climate change affects respiratory health, as well as outlining adaptation strategies. The latest statement provides an overview of all major pathways linking climate change with lung health.
While it summarises all of the available evidence, the authors also recognise some gaps in current knowledge. For instance, there is the need for further research to fully map the burden of climate change on respiratory diseases under different global warming scenarios and to understand underlying biological mechanisms, as well as identifying pathways of adaptation that can be translated into public health policies.
Professor Jovanovic Andersen, added: ‘Climate change affects everyone’s health, but arguably, respiratory patients are among the most vulnerable. These are people who already experience breathing difficulties and they are far more sensitive to our changing climate. Their symptoms will become worse, and for some this will be fatal.
‘Air pollution is already damaging our lungs. Now the effects of climate change are becoming a major threat to respiratory patients.’
Indeed, the deleterious respiratory effects of the particulate matter contained within air pollution, are already known to provide a mechanism through which lung cancer can develop among individuals who have never smoked.
Greater ethnic diversity in pioneering breast cancer clinical trials is the primary objective of a recent collaborative pilot project between Barts Health NHS Trust, The Christie NHS Foundation Trust, Roche Products Ltd and Macmillan Cancer Support.
The evidence derived from randomised controlled trials constitutes the most rigorous test of efficacy, effectiveness and safety for healthcare interventions. Nevertheless, if participants enrolled in these trials do not fully reflect the wider population for which the intervention is designed, the generalisability of the findings are limited.
What’s more, a 2016 study published in the British Journal of Cancer shows that young black women with breast cancer have more aggressive tumour profiles, present with later stages of disease, have higher mortality rates and experience poorer cancer care, yet they are often under-represented in clinical trials.
In an effort to redress this imbalance, this pilot project, which will run until August 2024, aims to increase the representation of black, Asian and ethnic minority women in breast cancer trials by improving access and identifying new and better ways to disseminate information.
This will include creating more targeted and meaningful communications for the communities the project is aiming to reach; increasing data, comparative baselines and patient retention records for research purposes; and providing enhanced support to ensure breast cancer patients understand the disease, what clinical research is and navigating patients to suitable clinical trials.
Findings and recommendations from the project will be used to create a case study and framework for future clinical trials and improve representation.
Dr Peter Hall, a medical oncology consultant at Barts Health NHS Trust who is involved in the project, said: ‘It’s well known that we need to do more to improve the diversity of participants taking part in the clinical trials we run at Barts Health and indeed, across the NHS. That’s why I’m so excited about this project.
‘By taking a targeted approach to driving diversity in clinical trials for a specific disease, we can not only improve representation for this condition, but learn how we can do this for trials into other conditions too. Ultimately, it will help us be more confident that the treatments we’re providing really do work for everyone. It’s also great to see this project being run as a collaboration between the NHS, charity and the commercial sector.‘
Understanding why clinical trials have historically under-represented people of different ethnicities remains unclear. Some reasons include the need for a narrowly defined, homogenous population to reduce variance and hence sample size, together with the imposition of stringent inclusion and exclusion criteria.
A further possible reason is the lack of engagement with ethnic communities. According to Charles Kwaku-Odoi, chief executive of the Caribbean African Health Network, ‘across the black community there is an undoubted legacy of disengagement in research and most certainly clinical trials that stems back decades as a result of mistrust. This has not served us well because it leads to a lack of appropriate interventions that perpetuate the grave health inequalities in breast cancer care‘.
He added: ‘This partnership approach to build solutions to improve engagement in clinical trials in breast cancer treatment and care is very much welcomed. We are looking forward to working in a collaborative way to build trust, improve awareness and ensure that barriers surrounding access to clinical trials are addressed.‘
In 2020, the INCLUDE group set out a multicomponent work stream project to improve representation of under-served groups in clinical trials. Under-representation of patients from different ethnicities is a recognised problem. For instance, research has revealed a lack of ethnic diversity within lipid lowering drug trials.
Two genes appear to be responsible for conferring chemoresistance in the majority of drug-resistant cell strains of patients with head and neck cancers, and silencing either gene leads to a complete reversal of drug resistance, researchers at Queen Mary University of London (QMUL) have found.
In the study, published in the journal Molecular Cancer, the team used transcriptome data-mining to identify potential genes that may be affecting tumour responsiveness to drug therapy. They identified a total of 28 genes in 12 strains of chemoresistant cell lines each against cisplatin, 5-fluorouracil, paclitaxel and docetaxel chemotherapies.
A total of 10 multi-drug chemoresistance genes were identified, four of which – TOP2A, DNMT1, INHBA and NEK2 – were up-regulated in a cohort of 221 head and neck cancer patients.
The INHBA and NEK2 genes appeared to be pan-cancer prognostic markers for predicting poor survival outcome in the majority of cancer types. But the team also identified two compounds – sirodesmin A and carfilzomib – from drug library screens, which were able to target both INHBA and NEK2 and re-sensitise cisplatin-resistant cells.
Dr Muy-Teck Teh, senior author of the study from QMUL, said: ‘These results are a promising step towards cancer patients in the future receiving personalised treatment based on their genes and tumour type that give them a better survival rate and treatment outcome.
‘Unfortunately, there are lots of people out there who do not respond to chemotherapy or radiation. But our study has shown that in head and neck cancers at least it is these two particular genes that could be behind this, which can then be targeted to fight against chemoresistance.‘
In Europe, head and neck cancers affect around 22 people per 100,000. While the cure rate is high for early-stage disease, around two-thirds of patients present with advanced-stage disease with a poor survival outcome. An additional and important cause of treatment failure leading to a poor survival, is the development of resistance to chemo and/or radiotherapy, although the underlying genes responsible for chemoresistance have previously been unclear.
4th September 2023
Delays in gut microbiome maturation in young children are uniformly associated with distinct allergic diagnoses at five years of age, according to the findings of a study by Canadian researchers.
The study, which was published in the journal Nature Communications, revealed how specific gut microbiome features and early life influences are associated with children developing any of four common allergies: atopic eczema, asthma, food allergy and/or allergic rhinitis.
It is possible, therefore, that these findings could lead to methods for predicting whether a child would develop an allergic disease. They could also form the foundation of strategies to prevent them from developing, especially given that food allergies in particular continue to be a major source of life-threatening reactions in children.
Courtney Hoskinson, PhD candidate at the University of British Columbia (UBC) and the study‘s lead author said: ‘Typically, our bodies tolerate the millions of bacteria living in our guts because they do so many good things for our health. Some of the ways we tolerate them are by keeping a strong barrier between them and our immune cells and by limiting inflammatory signals that would call those immune cells into action.‘
‘We found a common breakdown in these mechanisms in babies prior to the development of allergies.‘
In the study, researchers evaluated the four clinically distinct allergic diseases diagnosed at five years of age in the large, deeply characterised CHILD cohort study. The team adopted a multi-omics approach to profile infant stool collected at study visits scheduled for ages three months and one year.
The study used a deeply phenotyped cohort of 1,115 children. A total of 523 participants could be defined as a ‘healthy‘ control group in that they did not develop allergic sensitisation at any time in their life up to five years of age.
Some 592 children had been diagnosed by an expert physician at the five-year scheduled visit with one or more allergic disorders: atopic eczema (n = 367), asthma (n = 165), food allergy (n = 136) and allergic rhinitis (n = 187). There were gut microbiome features uniformly associated with these allergic diagnoses at five years of age.
When evaluating the association between early-life factors and a diagnosis of allergic disease at age five, male sex, a history of either maternal of paternal atopy and antibiotic usage before age one were all significantly linked with an increased risk of developing an allergic disease.
In contrast, breastfeeding up to age six months and self-identifying as Caucasian were negatively associated with an allergy diagnosis.
Dr Stuart Turvey, professor in the department of pediatrics at UBC, investigator at British Columbia Children‘s Hospital Research Institute and co-senior author on the study, added: ‘There are a lot of potential insights from this robust analysis. From these data we can see that factors such as antibiotic usage in the first year of life are more likely to result in later allergic disorders, while breastfeeding for the first six months is protective. This was universal to all the allergic disorders we studied.
‘Developing therapies that change these interactions during infancy may therefore prevent the development of all sorts of allergic diseases in childhood, which often last a lifetime.‘
High levels of two enzymes, CD39 and CD73, involved in the adenosine pathway are associated with reduced patient survival in early-stage non-small cell lung cancer (NSCLC) if found in tissue near the tumours, according to the findings of a new study.
Understanding the pathways of immune suppression is crucial to the development of new therapeutic targets for immunotherapy. In the study by the Centre for Inflammation Research at the University of Edinburgh and funded by the charity Cancer Research UK, researchers investigated why cancer immunotherapy fails so often by examining molecules that can interfere with the activity of T cells.
Published in the Journal for Immunotherapy of Cancer, the study revealed that higher levels of the CD39 and CD73 enzymes may help predict patient survival and indicate whether or not treatments will work. The researchers say the findings could help to pave the way for improved immunotherapies, allowing them to work more effectively in more patients.
NSCLC is the most common form of cancer, accounting for more than 80% of cases. While it is already known that T-cell subsets play a key role in shaping anticancer responses, there is still much to be learned to enable the development of diagnostic and therapeutic approaches.
The roles of the two enzymes, CD39 and CD73 have already been established. For instance, CD39 converts extracellular adenosine triphosphate to adenosine monophosphate and which is ultimately hydrolysed into adenosine by CD73. This liberated adenosine has highly immunosuppressive effects, enhancing the activity of suppressive immune cells.
For the study, the researchers turned to a cohort of 162 treatment naive early-stage NSCLC patients. They quantified the expression and localisation of CD39, CD73 and the tissue resident memory marker CD103.
They showed that in early, untreated NSCLC, patients had up-regulated expression of CD39 in the tumour tissue of natural killer cells, fibroblasts and T cells. In contrast, CD73 expression was mainly found among fibroblasts and Epcam+cells in the tumour tissue.
Further analysis revealed how CD39 expression was mainly localised in the tumour stroma, whereas CD73 expression was equally distributed between the tumour nest and stroma. Moreover, this high expression of both CD39 and CD73 in the tumour stroma was associated with poor recurrence-free survival at five years.
The team also observed that CD8+T cells within the tumour nest expressed CD103 and that the density of CD39+CD103+CD8+ T cells in the tumour nest, conferred a survival advantage, as witnessed by improved recurrence-free survival at five years.
Dr Ahsan Akram, Cancer Research UK clinician scientist fellow at the University of Edinburgh’s Centre for Inflammation Research, said: ‘This study helps us to understand that we need to know the types of T cells in the cancer and their location within the tumours to begin to appreciate the complexity we are dealing with.
‘We hope these results will lead to more research in this area, and in the future could help to identify patients who will do well with immunotherapies, as well as identifying earlier those that may not, so alternative treatments can be tried.‘
Further research and tests are needed alongside the integration of new technologies before any application in clinical practice is possible.
Individuals with a greater exercise capacity have a reduced risk of developing atrial fibrillation (AFib), ischaemic stroke and major adverse cardiovascular events (MACE), according to the findings of a large study presented at the European Society of Cardiology (ESC) Congress, 2023.
AFib is the most common cardiac arrhythmia and has a number of different causes including auto-immune diseases such as rheumatoid arthritis.
Whether being physically fit might reduce the risk of developing AFib is unclear, although some evidence reveals a graded, inverse relationship between cardiorespiratory fitness and incident AFib, especially among obese individuals.
The study included 15,450 individuals without AFib who had a mean age of 54.9 years (59% male). All participants were referred for a treadmill test between 2003 and 2012.
Fitness was assessed using the Bruce protocol, where participants are asked to walk faster and at a steeper grade in successive three-minute stages. It was then calculated according to the rate of energy expenditure the participants achieved, which was expressed in metabolic equivalents (METs).
Participants were then divided into three fitness levels according to the METs achieved during the treadmill test: low (less than 8.57 METs), medium (8.57 to 10.72) and high (more than 10.72).
The researchers looked for independent associations between exercise capacity on the treadmill and the risk of new-onset AFib, risk of ischaemic stroke and MACE. The results were adjusted for potential confounders including age, sex, cholesterol level, kidney function, prior stroke, hypertension and any medications.
During the period of follow-up, new-onset AFib occurred in 3.33% of participants.
In fully adjusted models, each one MET increase in exercise treadmill testing, there was an associated 8% lower risk of AFib incidence (hazard ratio, HR = 0.92, 95% CI 0.88 – 0.97).
In addition, this one MET increase was also associated with a lower risk of ischaemic stroke (HR = 0.88, 95% CI 0.83 – 0.94) and a 14% reduced risk of MACE (HR = 0.86, 95% CI 0.84 – 0.88).
In fact, the probability of remaining free from AFib over a five-year period was calculated to be 97.1%, 98.4% and 98.4% in the low, medium and high exercise capacity groups, respectively.
Study author Dr Shih-Hsien Sung of the National Yang Ming Chiao Tung University in Taipei, Taiwan, said: ‘This was a large study with an objective measurement of fitness and more than 11 years of follow up. The findings indicate that keeping fit may help prevent atrial fibrillation and stroke.‘
1st September 2023
With lack of time often cited as a barrier to undertaking physical activity, cramming a week‘s worth of exercise into a day or two may seem more achievable, especially if it provides comparable cardiovascular benefits to a more evenly distributed pattern. Hospital Healthcare Europe‘s clinical writer and resident pharmacist Rod Tucker considers the evidence.
It is abundantly clear that being physically active is associated with health benefits. Current guidance on physical activity for adults in most major countries in Europe is broadly similar: undertake either at least 150 minutes of moderate intensity activity a week or 75 minutes of vigorous intensity activity, plus two days a week of strengthening activities to work all of the main muscle groups.
Moreover, it is advocated that exercise is undertaken every day or spread evenly over four to five days a week.
But is there any evidence that achieving this amount of exercise is associated with health benefits? In a 2020 study, an international research group tried to answer this question.
The team looked at the association between attainment of the recommended amount of physical activity among a representative sample of US citizens, and all-cause and cause specific mortality.
Their analysis included 479,856 adults who were followed for a median of 8.75 years. The findings were very clear: undertaking the recommended amounts of physical activity reduced the risk of all-cause mortality by 40%. But not only that, such levels of activity lowered the risk of cardiovascular disease by 50% and the risk of cancer by 40%.
With clear evidence of the health benefits from undertaking the recommended levels of physical activity, surveys have also identified some notable and recognised barriers to exercising. One of the most consistently reported barriers is sufficient time to exercise, and this is seen irrespective of age and gender.
Although healthcare professionals may advocate that their patients engage in exercise as part of the health promotion message, it seems they don‘t always practise what they preach. In fact, research shows that lack of time is also a perceived barrier to exercise among doctors and nurses.
As well as insufficient time, a demanding workload gives rise to high levels of burnout and stress, and changing shift patterns can limit time and motivation, which all represent additional barriers to exercising among clinicians.
Evidence suggests that exercising for just one or two days a week could accrue the same health benefits seen among those who exercise more regularly throughout the week. For example, so-called ‘weekend warriors‘ – those who restrict physical activity to just one or two sessions per week – have a similar level of all-cause mortality compared to those who spread their physical activity over several days.
In fact, a 2023 meta-analysis of four studies with 426,428 participants found that the risk of both cardiovascular disease mortality and all-cause mortality in those compressing their activity into two days was 27% and 17% lower, respectively, when compared to those who were inactive.
However, a limitation of this analysis was that levels of physical activity were self-reported and therefore prone to misclassification bias.
With inherent self-reporting bias an issue, a recent study examined the value of accelerometer-derived data. Researchers recently set out to examine the association between a weekend warrior pattern of moderate-to-vigorous physical activity (MVPA) achieved over just one to two days, compared to the activity being spread more evenly, with the risk of incident cardiovascular events.
The researchers retrospectively analysed a UK Biobank cohort who provided a full week‘s worth of wrist-based accelerometer physical activity data. Individuals were classified into three groups: active weekend warriors, in which more than half of their total MVPA was undertaken over one to two days; regularly active, where exercising was spread throughout the week; and inactive, where less than 150 minutes of MVPA per week was undertaken.
They looked at associations between the different activity pattern and cardiovascular outcomes such as incident atrial fibrillation, myocardial infarction, heart failure and stroke. The findings were then adjusted for several potential covariates including age, sex, ethnicity race, alcohol and smoking and diet quality.
Data for 89,573 individuals with a mean of 62 years (56% female) were included in the analysis and who were followed for a median of 6.3 years. Interestingly, when stratified at the threshold of 150 minutes or more of MVPA per week, nearly half of the entire cohort (42.2%) were classed as weekend warriors.
The findings for atrial fibrillation, myocardial infarction, heart failure and stroke are summarised in the table below.
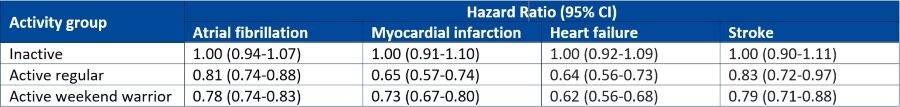
The accelerometer-derived study shows that engagement in physical activity, regardless of the pattern, is able to reduce the risk of a broad range of adverse cardiovascular outcomes. Therefore, although many healthcare professionals‘ working weeks may not permit specific exercising days, it seems that compressing physical activity into just two days per week, wherever possible, still achieves comparable health benefits.
So, whether it’s advice for patients or the foundation for personal fitness goals, the key message is to just keep moving.
30th August 2023
Young men with a higher level of cardiorespiratory fitness have a significantly lower risk of developing several cancers in later life, according to the findings of a new study published in the British Journal of Sports Medicine.
It is already known that aerobic exercise induces interleukin-6 and suppresses a marker of DNA damage, which may account for a protective role in colon cancer. But whether being fit could reduce the risk of developing cancer in later life is far less clear.
For the recent study, Swedish researchers set out to assess the associations between cardiorespiratory fitness in young men and the incidence of site-specific cancer.
They turned to data held on men who underwent military conscription between 1968 and 2005 and for whom cardiorespiratory function was assessed by maximal aerobic workload cycle test at conscription.
The men’s level of fitness was then categorised as low, moderate or high, and those who received a cancer diagnosis before or within five years after the military conscription were excluded from the analysis.
The team included 1,078,000 men, of whom 6.9% subsequently developed cancer in at least one site during a mean follow-up of 33 years.
A higher cardiorespiratory fitness was linearly associated with a significantly lower risk of developing nine different cancers. This included cancer in the head and neck (Hazard ratio, HR = 0.81), oesophagus (HR = 0.61), stomach (HR = 0.79), pancreas (HR = 0.88) and liver (HR = 0.60).
In contrast, a higher cardiorespiratory fitness significantly increased the risk of being diagnosed with prostate cancer (HR = 1.07) and malignant skin cancer (HR = 1.31).
While it is an observational study and no firm conclusions can be drawn about cause and effect, the researchers suggested that the findings strengthened the incentive for promoting interventions aimed at increasing cardiorespiratory fitness in younger people.
The use of intravenous ferric carboxymaltose (FCM) in heart failure patients with iron deficiency reduces the risk of hospitalisation and cardiovascular death, according to research presented at the recent European Society for Cardiology (ESC) Congress 2023 in Amsterdam.
Researchers undertook a meta-analysis of individual participant data from three randomised, placebo-controlled trials of FCM in adult patients with heart failure and iron deficiency: CONFIRM-HF, AFFIRM-HF and HEART-FID.
Across the three trials, a total of 4,501 patients with heart failure and reduced or mildly reduced left ventricular ejection fraction and iron deficiency were randomly assigned to FCM (n = 2,251) or placebo (n=2,250) for 52 weeks. The mean age of the total population was 69 years, 63% were men and the mean left ventricular ejection fraction was 32%.
Researchers set the primary efficacy endpoints as a composite of total cardiovascular hospitalisations and cardiovascular death, as well as a composite of total heart failure hospitalisations and cardiovascular death. Key secondary endpoints included individual components of the composite endpoints.
The trial results revealed FCM therapy significantly reduced the co-primary composite endpoint of total cardiovascular hospitalisations and cardiovascular death compared with the placebo (rate ratio, RR = 0.86, 95% CI 0.75 – 0.98, p = 0.029).
Although there was a trend towards reduction of the co-primary composite endpoint of total heart failure hospitalisations and cardiovascular death, this was not statistically significant (RR = 0.87, 95% CI 0.75 b- 1.02, P = 0.076).
Nevertheless, FCM therapy was associated with a 17% relative rate reduction in total cardiovascular hospitalisations (RR = 0.83, 95% CI 0.73 – 0.96, p = 0.009) and a 16% relative rate reduction in total heart failure hospitalisations (RR = 0.84, 95% CI 0.71 – 0.98 p = 0.025). Despite these benefits, FCM therapy had no effect on mortality.
Professor Piotr Ponikowski, the principal investigator and vice-rector of Wroclaw Medical University, Poland, said: ‘This was the largest and most up-to-date analysis of the effect of FCM in iron-deficient heart failure patients with reduced or mildly reduced ejection fraction.‘
He added: ‘The findings indicate that intravenous FCM should be considered in iron-deficient patients with heart failure and reduced or mildly reduced ejection fraction to reduce the risk of hospitalisation due to heart failure and cardiovascular causes.‘
Heart failure is one of the leading causes of avoidable hospitalisations and iron deficiency is present in over 30% to 50% of patients.
Although iron therapy is known to improve functional capacity, symptoms, and quality of life, until the current meta-analysis, no studies have examined whether treatment impacts on clinical events such as hospitalisation.