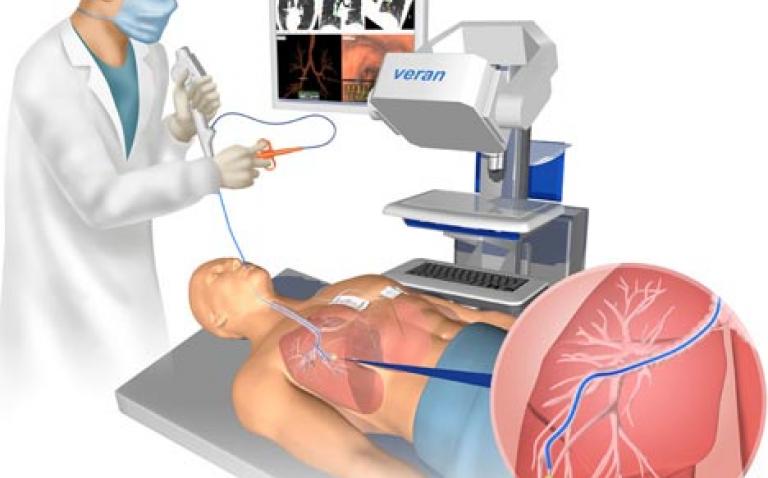
Veran Medical Technologies announced that it has recently received two clearances (K093146 & K093995) from the U.S. Food and Drug Administration (FDA) to expand its indications for use of the ig4 Navigation platform.
The addition of the Ultrasound and 3D Fluoroscopic X-ray image modalities represent an important advancement for the ig4 Navigation platform.
The ability to perform navigated cancer thermal ablation in lower cost imaging suites greatly increases the access and cost effectiveness of these procedures while maintaining the high-resolution image quality provided by conventional CT guidance.
“History has shown that the delivery of therapies in a cost effective, reliable, and efficient manner is as important as the therapy itself. We are excited about the near future when we will have tightly integrated therapy and delivery as well. These clearances allow Veran to be a market-unlocking key in driving the growth of minimally invasive interventional oncology procedures,” said Les Carlson, Veran Director of Percutaneous Products.