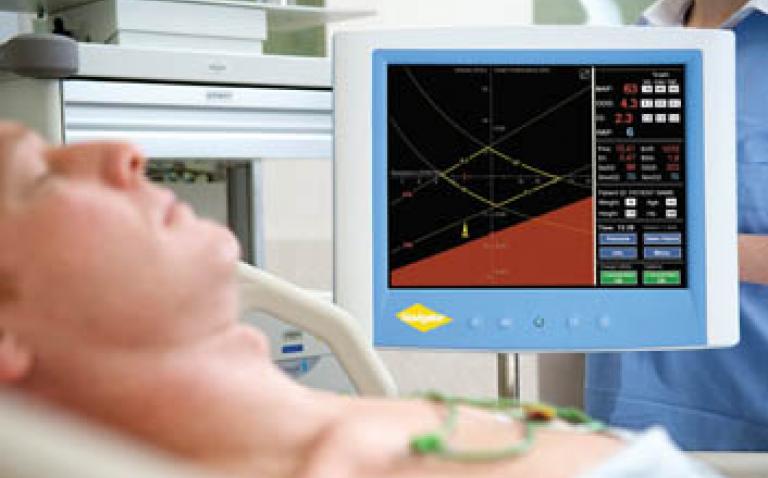
Navigator™ provides a complete picture of the cardiovascular system for clinical decision making. It enables a systematic approach to cardiovascular management and provides real time, dynamic and integrated interpretation of patient measurements, and the effects of treatments on them.
Philips Healthcare is the leading supplier in the critical care patient monitoring market. Its new range of MX patient monitors contains an integrated PC option which enable it to run a range of clinical applications within the monitor.
Mark Leaning, Executive Director at Applied Physiology, said:
“The primary objective of this pilot study was to assess customer satisfaction levels in the areas of clinical utility, perceived user value and desire to purchase Applied Physiology’s Navigator™. The feedback has been very positive with clinicians all reporting that our software improved patient care, that they were likely to recommend Navigator™ to a friend or colleague, and would be interested in purchasing Navigator for continued use. As a result of the successful trial Navigator™ is now embedded in a Philips MX800 monitor at Monash Medical Centre in Melbourne, Australia. Feedback on this will be shared in a few months.”
A spokesperson at Philips Healthcare said:
“A key element of our strategy is for Philips to be able to offer next generation products that take current measured data and transform it into useful information for patient assessment and treatment decision-making. Clinical decision support systems – such as Applied Physiology’s Navigator™ – are an excellent example of this type of cutting edge technology.”
All respondents reported that overall the Navigator™ assisted with patient care either ‘substantially’ (40%) or ‘substantially/moderately’ (60%). Users reported that Navigator™ influenced patient care through better fluid management (5/5 respondents), titration of noradrenaline (5/5 respondents) and clarification of cardiovascular targets (5/5 respondents). The study commenced in September 2011, and concluded in August 2012. Five users from three sites took part in the interviews. Their roles were senior intensivist, cardiology resident, consultant in critical care, director of critical care services and staff specialist consultant.