A Swiss pharmaceutical and diagnostics company is launching a fully-automated immunoassay for the determination of autoantibodies to cyclic citrullinated peptides (anti-CCP).
The assay, produced by Roche Diagnostics, has been CE approved for use on the Elecsys/cobas e electrochemiluminescence immunoassay systems. The results of the anti-CCP assay are intended to be used as an aid in the diagnosis of rheumatoid arthritis, in combination with other clinical and laboratory findings.
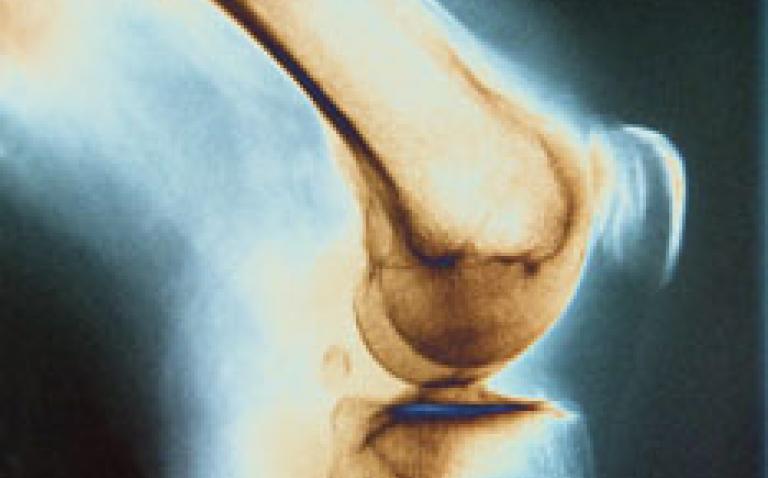
Rheumatoid arthritis is one of the most common autoimmune diseases, affecting 0.5–1% of the world’s population. This systemic disease is characterised by chronic inflammation of the synovial joints and progressive joint degeneration leading to loss of movement and to deformity over time.
Early diagnosis of rheumatoid arthritis is important as early treatment may help to prevent joint damage and subsequent disability.
The diagnosis of rheumatoid arthritis relies on clinical symptoms and laboratory tests, such as rheumatoid factor and C-reactive protein. Anti-CCP is superior to both parameters in terms of diagnostic specificity and predicts more severe disease course and outcome, according to Roche.
“We believe the Elecsys Anti-CCP plays an important role in the early diagnosis and treatment of rheumatoid arthritis and provides an excellent tool for primary care physicians and rheumatologists,” said Dirk Ehlers, Head of Roche Professional Diagnostics.
The Elecsys anti-CCP test marks another successful step in our strategy of offering the most comprehensive immunoassay test menu.”