St. Jude Medical Inc has announced the Nanostim™ leadless pacemaker was awarded the 2014 Innovation Award at Cardiostim 2014, the 19th World Congress in Cardiac Electrophysiology and Cardiac Techniques.
The award, which was chosen by a congress committee of electrophysiologists and conveys the support of the electrophysiology (EP) community, recognises the Nanostim leadless pacemaker as the industry’s most innovative product in the sector of electrophysiology and cardiac techniques.
“St. Jude Medical is honoured that our Nanostim pacemaker received this significant recognition from the EP community,” said Eric S Fain MD, group president of St. Jude Medical. “With more than 55 years of history in pacing innovation, we are proud that this revolutionary technology is transforming the treatment of cardiac arrhythmias and setting a new standard of care for future pacing technologies. We believe that the health benefits associated with this leadless technology will enable physicians who use this device to improve the lives of patients worldwide.”
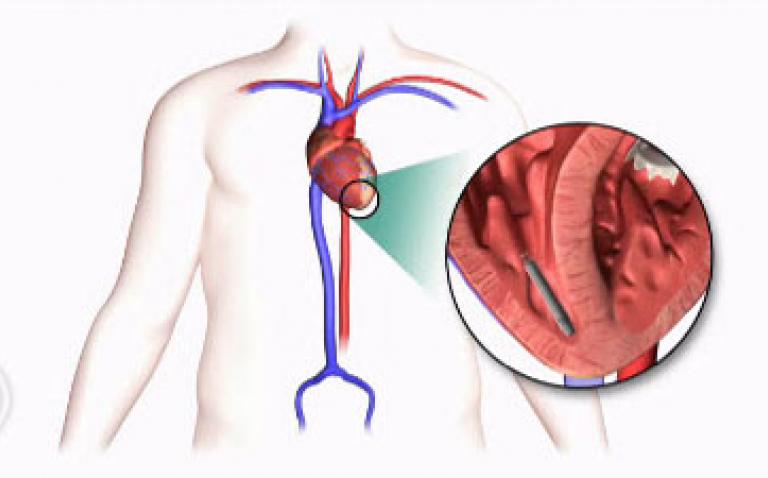
More than 4 million people worldwide have an implanted pacemaker or other cardiac rhythm management device, and an additional 700,000 patients receive the devices each year. Unlike conventional pacemakers that require a more invasive surgery, the Nanostim leadless pacemaker is designed to be implanted directly into the heart via a non-surgical procedure.
The device, which earned CE Mark approval in 2013, is designed to be placed without the visible surgical pocket, scar and insulated wires (called leads) required for conventional pacemakers. Implanted via the femoral vein with the smallest available leadless technology delivery system (includes the delivery system catheter and the Nanostim™ 18 F introducer), the device is designed to be fully retrievable so that it can be readily repositioned throughout the implant procedure and later retrieved if necessary.
The Nanostim leadless pacemaker is less than 10% the size of a conventional pacemaker and is the least invasive pacing technology on the market today. The small size of the device and lack of a surgical pocket, coupled with the exclusion of a lead, improves patient comfort and can reduce traditional pacemaker complications, including device pocket-related infection and lead failure. The device battery is expected to have an average lifespan of more than nine years at 100% pacing, or more than 13 years at 50% pacing.
The elimination of the visible lump and scar at a conventional pacemaker’s implant site, in addition to the removal of patient activity restrictions that may prevent the dislodgement or damage to a conventional lead, will potentially improve the quality of life for patients with this technology by allowing most to continue living active, uninhibited lifestyles. The device is supported by the St. Jude Medical Merlin™ Programmer, which is also used to interrogate and program the company’s other pacemakers and implantable cardioverter defibrillators (ICDs).
Cardiac pacemakers are used to treat bradycardia, which is a heart rate that is too slow. These devices monitor the heart and provide electrical stimulation when the heart beats too slowly for each patient’s specific physiological requirements.
For more information about Cardiostim 2014 and St. Jude Medical, visit: www.sjm.com/cardiostim.